* The electronic layout shown in the above virtual experiment is based on Wikipedia data. Similarly, before the 4d orbital is filled with electrons, it is first filled in the 5s orbital.īesides, electrons have the property of being in pairs (even) as can as possible.Īlso, there are cases where electrons become stable as they fill the upper orbital first. So, before the 3d orbital is filled with electrons, the 4s orbital is filled with electrons first. Groups one and two are the s-block, three through 12 represent the d-block, 13 to 18 are the p-block and the two rows at the bottom are the f-block. To calculate an electron configuration, divide the periodic table into sections to represent the atomic orbitals, the regions where electrons are contained. When the number of shells exceeds 3, the energy boundary begins to become unclear.įor example, a 4s orbital has a lower energy value compared to a 3d orbital. Electron configurations help you to do this. In other words, as the energy increases in the order of s → p → d → f, the energy level increases. 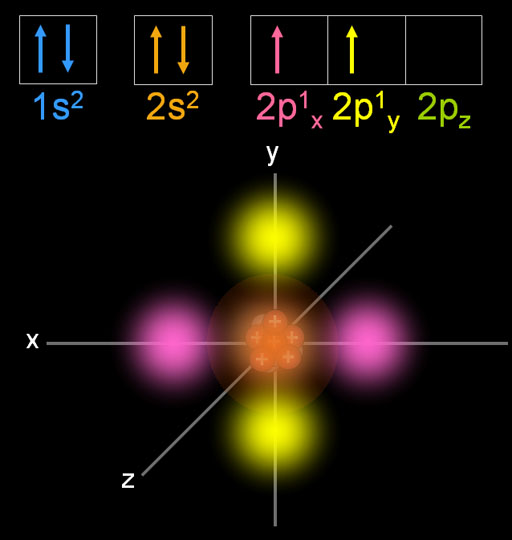
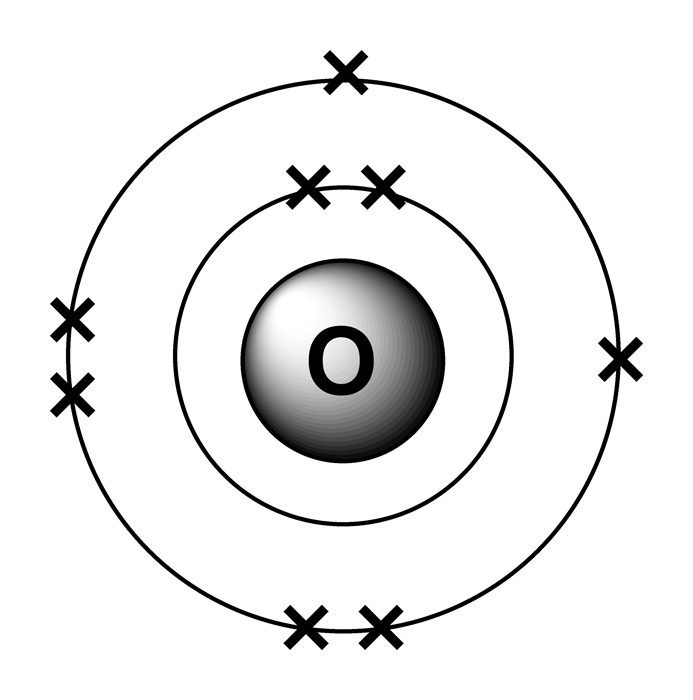
The first is electron configuration, which is directly related to an elements position. In the same shell, electrons are filled in the order of s, p, d, f orbitals. Many atomic properties can be related to position on the periodic table. In a stable atom, electrons are filled from the innermost shell. If both the s orbital and the p orbital of the same shell are empty, the electrons first try to fill the s orbital first. The s orbital has the lowest energy so that the electrons can be the most stable. From the fourth shell (n>=4) there are s, p, d, f orbitals.Įach orbital has a fixed number of electrons.Įach orbital has a slightly different electrical potential energy.The third shell (n=3) has s, p, d orbitals.The atomic number (or proton number) is 8. First, go to the periodic table and find how many electrons oxygen has. Use the periodic table to write condensed electron configurations and. We need to write the electron configuration for an oxygen atom. The second shell (n=2) has an s orbital and a p orbital. Draw an energy-level diagram for the orbitals in a many-electron atom and.The innermost shell (n=1) only has an s orbital.Type of orbitalĮach shell in an atom has a different type of orbital. In an atom, electrons form a ‘shell (layer),’ and each shell comprises several types of orbitals. An atom has an atomic nucleus with a (+) charge at the center and electrons with a negative charge around it.Įlectrons are arranged according to certain rules (wave functions), which are called ‘orbitals.’
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
